Topic 5: Nuclear receptors in circadian biology
Presentation of research topics
The biological clock plays an important physiological role. For example, it generates circadian rhythms in sleep, blood pressure, immune response, and metabolism. Disruption of the clock—through shift work, frequent jet lag, or exposure to light at night—increases the risk of developing metabolic diseases (obesity, type 2 diabetes, NAFLD), as well as inflammatory and cardiovascular diseases (atherosclerosis, myocardial infarction).
By working on clock components and the nuclear receptors Rev-erbs and RORs, we aim to understand the cellular and molecular mechanisms by which the clock controls metabolism and inflammation in various pathophysiological contexts such as diabetes, MASLD, atherosclerosis, and muscle regeneration. We seek to determine whether and how the pharmacological modulation of these nuclear receptors can restore circadian rhythmicity and improve these pathological conditions.
Our team is engaged in collaborations with other teams within UMR1011, as well as with regional, national, and international teams, in order to complement our expertise and move toward translational studies
List of key words :
Circadian rythms ; Metabolism ; Inflammation ; Récepteurs nucléaires ; Cardiovascular and metabolic diseases
Dr Helene Duez, PhD, HDR
Research director 1, INSERM
helene.duez[@]pasteur-lille.fr
ORCID : 0000-0002-4130-7987
I lead the team “Circadian Rhythms in Cardiometabolic and Inflammatory Diseases” (UMR1011) in Lille. During my PhD at the Institut Pasteur de Lille (defended in 2003), I identified the role of nuclear receptors in lipid and bile acid metabolism, as well as in the development of atherosclerosis. I then completed a postdoctoral fellowship at the University of Toronto, where I discovered that the production of triglyceride-rich lipoproteins by the intestine is increased in insulin-resistant patients. My research has also focused on the role of incretins in glucose metabolism.Recruited as a CRCN at Inserm in 2008, I have since led a research team dedicated to studying the cellular and molecular mechanisms by which the biological clock influences metabolism and inflammation in various pathophysiological contexts, including metabolic, cardiovascular, and muscle diseases.
I have received numerous competitive grants, including a European MSCA ReIntegration Grant, funding from the French National Research Agency (ANR), the FRM “Équipe” program, the European Foundation for the Study of Diabetes (EFSD), the Fondation Francophone pour la Recherche sur le Diabète (FFRD), the Société Francophone du Diabète (SFD), and the CPER “Équipe émergente” program. I have contributed to more than 60 peer-reviewed publications in leading journals such as Nature Medicine, Gastroenterology, The Lancet, Nature Cardiovascular Research, Journal of Clinical Investigation, and Circulation, and have delivered more than 70 invited lectures.
I serve on several evaluation committees, including the ERC Starting Grant LS4 panel, the scientific boards of the FFRD and FRM, as well as various scientific advisory boards (SABs) and international expert committees.
Dr Benoit Pourcet, PhD, HDR
Assistant professor, Lille University
ORCID : 0000-0002-6758-5467
Benoit Pourcet, a biochemist by training, has been an Associate Professor at the Faculty of Pharmacy of the University of Lille since 2016. His research primarily focuses on the role and regulation of nuclear receptors in the context of cardiometabolic diseases. Since 2004, during his PhD at the Institut Pasteur de Lille and his postdoctoral training at University College London (United Kingdom), he has acquired strong expertise in the molecular mechanisms involved in the control of nuclear receptor transcriptional activity in the pathophysiological context of inflammatory, metabolic, and cardiovascular diseases.
He now leads a research group investigating the impact of the nuclear receptor Rev-erb-α on the development of pathologies induced by circadian clock disruption, such as atherosclerosis. This innovative research area complements the projects developed within Team 5 and Unit 1011.
Pr Yasmine Sebti, PhD, HDR
Professor, Lille University
ORCID : 0000-0002-1813-810X
Yasmine Sebti was recruited as an Associate Professor at the Faculty of Pharmacy of the University of Lille in 2014 and was promoted to Full Professor in September 2025. During her PhD at the University of Rennes and her postdoctoral training at the University of Oxford (United Kingdom), she developed strong expertise in immunology.
Since 2009, she has been a member of UMR1011, where she has developed research projects focused on the metabolism and pathophysiology of skeletal muscle. She currently leads a research group investigating the role of the biological clock—through the study of the nuclear receptors Rev-erbα and RORα—in inflammatory processes during muscle regeneration, thereby integrating the diverse expertise she has acquired throughout her career.
To support her research, she has secured competitive funding, including grants from the French National Research Agency (ANR), the Société Francophone du Diabète (SFD), and AFM-Téléthon.

Stephane Delhaye,
Engineer - Lille University
stephane.delhaye[@]univ-lille.fr
ORCID :0000-0002-1813-810X
Stéphane Delhaye obtained a professional Master’s degree in 2005. At the end of his studies, he first joined a laboratory at the Center for Infection and Immunity of Lille, before joining the INSERM Unit UMR1011 in 2012 in Dr. Hélène Duez’s team. In 2017, he was appointed as an Assistant Engineer within the team, before being promoted to Research Engineer in 2026. He is particularly involved in the development and use of experimental models for the team’s various research projects.
Christian Duhem,
Engineer - Institut Pasteur of Lille
christian.duhem[@]pasteur-lille.fr
ORCID : 0000-0003-0931-3815
Sarah Ampen,
Engineer, INSERM
ORCID : 0009-0009-0284-1763
Sarah Ampen is an engineer within Unit 1011, working under the supervision of Yasmine Sebti. She holds a Master’s degree in Omics and Systems Biology and develops approaches combining experimental biology and bioinformatics analyses. Her research focuses on the role of type 2 innate lymphoid cells (ILC2s) in skeletal muscle homeostasis. She investigates their molecular signature in regenerative and degenerative contexts, particularly in Duchenne muscular dystrophy, using histological analyses and transcriptomic approaches (RNA-seq, single-cell RNA-seq), in order to better understand their involvement in the regulation of muscle fibrosis.
Dr Lise Ferri, PhD
Postdoctoral researcher, INSERM
ORCID : 0000-0003-1981-4250
Lise Ferri is a postdoctoral researcher in physiology. Her work focuses on the impact of circadian rhythm disruption on the development of atherosclerosis. During her PhD, supervised by Dr. Benoit Pourcet, her research specifically investigated the role of the nuclear receptor Rev-erbα in the mechanisms leading to plaque instability, including vascular calcification and intraplaque neovascularization.
Margaux Leduc,
PhD Student, Lille University
margaux.leduc[@]pasteur-lille.fr
ORCID : 0009-0009-7432-9574
Margaux Leduc is a PhD student under the supervision of Prof. Yasmine Sebti and Dr. Hélène Duez. She joined Team 5 of Unit U1011 during her Master’s (M2) internship in Preclinical Experimentation Management. Her doctoral research focuses on evaluating the role of the biological clock in inflammatory processes during muscle regeneration.
Salomé Farge
PhD Student, Lille University
ORCID : 0009-0002-6366-3197
After completing her engineering degree, Salomé Farge joined Team 5 at U1001 to undertake her PhD under the supervision of Dr Benoit Pourcet. Her work focuses primarily on the role of the nuclear receptor Rev-erb-α in the mechanisms leading to plaque instability, in particular intraplaque neovascularisation caused by disrupted blood flow.
Recent works of the team :
- We have shown that Rev-erbα plays a central role in muscle physiology by regulating mitochondrial biogenesis and function (Woldt, Sebti et al., Nature Medicine 2013) and calcium fluxes (Boulinguiez, Duhem et al., JCI Insight 2022).
- We have discovered that susceptibility to the development of acute inflammatory conditions varies throughout the day and that Rev-erbα is a major player in circadian immunity by regulating the NLRP3 inflammasome (Pourcet et al., Gastroenterology, 2018)
- In a collaborative study, we demonstrated the impact of chronobiology on the incidence of post-operative cardiac conditions (Montaigne et al. The LANCET, 2018)
- We have demonstrated that insulin signalling is necessary for the circadian organisation of hepatic metabolism (Fougeray et al., Cell Reports 2022) and the role of the macrophage clock in the development of NASL in insulin-resistant obese mice (Hebras et al., in review).
- We have shown that the response to pneumococcal infection varies depending on the time of infection, and that this diurnal response is impaired in aged mice. Pharmacological modulation of Rev-erbα in aged mice improves their resistance to infection (Silva Angulo et al., Cell Reports 2025)
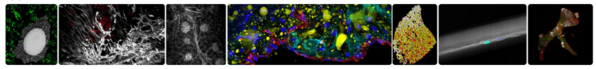
Team 5 of UMR1011 has developed expertise in a range of areas :
-We have developed a Fiji macro, MUSCLE J (Mayeuf-Louchart et al., Skeletal Muscle, 2018), which enables automated quantification of muscle fiber cross-sectional area, as well as the number and spatial distribution of nuclei, muscle stem cells, and endothelial cells.
-We have also established genetic and environmental models of circadian clock disruption to better understand the role of circadian regulators in the development of metabolic, inflammatory, and cardiovascular diseases.
-In addition, we have implemented organ clearing techniques combined with light-sheet microscopy and 3D image reconstruction, allowing comprehensive visualization of tissues at the whole-organ level.
-Our expertise also includes the following areas: chronobiology and chronopharmacology, metabolic phenotyping, mitochondrial respiration analysis using oxygraphy (Oroboros, Seahorse) and calcium flux measurements, immunophenotyping (FACS, immunohistochemistry), imaging (confocal microscopy, light-sheet microscopy, whole-organ imaging), RNAscope, and cell culture (cell lines and primary cells)
List of main publications from team 5 :
Woldt E. *, Sebti Y. *, Solt L.A., Duhem C., Lancel S., Eeckhoute J., Hesselink M.K.C., Paquet C., Delhaye S., Shin Y., Kamenecka T.M., Schaart G., Lefebvre P., Nevière R., Burris T.P., Schrauwen P., Staels B., Duez H. Rev-erb-α modulates skeletal muscle oxidative capacity by regulating mitochondrial biogenesis and autophagy. Nature Medicine, 2013, 19(8) : 1039-1046.
Montaigne David, Maréchal Xavier, Modine Thomas, Coisne Augustin, Mouton Stéphanie, Fayad Georges, Ninni Sandro, Klein Cédric, Ortmans Staniel, Seunes Claire, Potelle Charlotte, Berthier Alexandre, Gheeraert Celine, Piveteau Catherine, Deprez Rebecca, Eeckhoute Jérome, Duez Hélène, Lacroix Dominique, Deprez Benoit, Jegou Bruno, Koussa Mohamed, Edme Jean-Louis, Lefebvre Philippe, Staels Bart. Day-time variation of peri-operative myocardial injury in cardiac surgery and its prevention by Rev-erbɑ antagonism: a single-centre propensity-matched cohort study and a randomised study. The LANCET, 2018, 391:59-62
Lancel S, Hesselink MK, Woldt E, Rouillé Y, Dorchies E, Delhaye S, Duhem C, Thorel Q, Mayeuf-Louchart A, Pourcet B, Montel V, Schaart G, Breton N, Picquet F, Briand O, Salles JP, Duez H, Schrauwen P, Bastide B, Bailleul B, Staels B, Sebti Y. Endospanin-2 enhances skeletal muscle energy metabolism and running endurance capacity. JCI Insight, 2018, 3(9). pii: 98081. doi: 10.1172/jci.insight.98081
Pourcet B*, Zecchin M*, Ferri L, Beauchamp J, Sitaula S, Billon C, Delhaye S, Vanhoutte J, Mayeuf-Louchart A, Thorel Q, Haas J, Eeckhoute J, Dombrowicz D, Duhem C, Boulinguiez A, Lancel S, Sebti Y, Burris T, Staels B and Duez H. Nuclear Receptor Subfamily 1 Group D Member 1 (Rev-erb-α) Regulates Circadian Activity Of NLRP3 Inflammasome to Reduce the Severity of Fulminant Hepatitis in Mice. Gastroenterology, 2018, 154(5):1449-1464.e20
Mayeuf-Louchart A, Lancel S, Sebti Y, Pourcet B, Loyens A, Delhaye S, Duhem C, Beauchamp J, Ferri L, Thorel Q, Boulinguiez A, Zecchin M, Dubois-Chevalier J, Eeckhoute J, Vaughn LT, Roach PJ, Dani C, Pederson BA, Vincent SD, Staels B, Duez H. Glycogen Dynamics Drives Lipid Droplet Biogenesis during Brown Adipocyte Differentiation. Cell Rep. 2019, 29(6):1410-1418.e6. doi: 10.1016/j.celrep.2019.09.073. PMID: 31693883
Boulinguiez A*, Duhem C*, Mayeuf-Louchart A, Pourcet B, Sebti Y, Kondratska K, Montel V, Delhaye S, Thorel Q, Beauchamp J, Hebras H, Gimenez M, Couvelaere M, Zecchin M, Ferri L, Prevarskaya N, Forand A, Gentil C, Ohana J, Piétri-Rouxel F, Bastide B, Staels B, Duez H*, Lancel S*. Nuclear Receptor Subfamily 1 Group D Member 1 controls skeletal muscle calcium homeostasis through myoregulin repression. JCI Insight 2022 Sep 8;7(17):e153584. doi: 10.1172/jci.insight.153584.
Fougeray T, Polizzi A, Régnier M, Fougerat A, Ellero-Simatos S, Lippi Y, Smati S, Lasserre F, Tramunt B, Huillet M, Dopavogui L, Salvi J, Nédélec E, Gigot V, Smith L, Naylies C, Sommer C, Haas JT, Wahli W, Duez H, Gourdy P, Gamet-Payrastre L, Benani A, Burnol AF, Loiseau N, Postic C, Montagner A*, Guillou H*. The hepatocyte insulin receptor is required to program the liver clock rhythmic gene expression Cell Reports 2022, 39
Manjula Vinod, Alexandre Berthier, Xavier Maréchal, Céline Gheeraert, Raphaël Boutry, Stéphane Delhaye, Jean-Sébastien Annicotte, Hélène Duez, Agnès Hovasse, Sarah Cianférani, David Montaigne, Jérôme Eeckhoute, Bart Staels, Philippe Lefebvre. Timed use of digoxin prevents heart ischemia-reperfusion injury through a REV-ERBα-UPS signaling pathway. Nature Cardiovascular Research, 2022, 1, 990-1005.
J. Yang, H.D. de Vries, A. Mayeuf-Louchart, J.H. Stroeve, V.W. Bloks, M. Koehorst, H. Duez, B. Staels, F. Kuipers, T. van Zutphen. Role of bile acid receptor FXR in development and function of brown adipose tissue. Biochim Biophys Acta - Molecular and Cell Biology of Lipids. 2023;1868(2):159257.
Anne Danckaert*, Aurélie Trignol, Guillaume Le Loher, Sébastien Loubens, Bart Staels, Hélène Duez, Spencer L. Shorte, Alicia Mayeuf-Louchart*. MuscleJ2: a rebuilding of MuscleJ with new features for high‑content analysis of skeletal muscle immunofluorescence slides. Skeletal Muscle 2023;13(1):14
F. Silva Angulo, C. V. Joseph, L. Delval, L. Deruyter, S. Heumel, M. Bicharel, P. Brito Rodrigues, V. Sencio, T. Bourguignon, M. Gomes Machado, M. Fourcot, S. Delhaye, S. Salomé-Desnoulez, P. Valet, S. Adnot, I. Wolowczuk, J. C. Sirard, M. Pichavant, B. Staels, J. T. Haas, R. Gref, J. Vandel, A. Machelart, H. Duez$, B. Pourcet*$, and F. Trottein*$. Rev-erb-α antagonism in alveolar macrophages protects against pneumococcal infection in elderly mice. Cell Reports 2025, Volume 44, Issue 2, 115273
List of main fundings from team 5 :
Fondation pour la Recherche Médicale (FRM), Agence Nationale pour la Recherche (ANR), ANR-Labex-EGID, AFM-téléthon, Fondation de France, Société Française du Diabète, Fédération Française de Cardiologie, Institut Pasteur de Lille ; Université de Lille ; Inserm
Dr Duez Hélène
Research director, INSERM,
INSERM U1011 – Institut Pasteur de Lille-Université Lille Nord de France – EGID
1 rue Calmette, BP245, 59019 Lille
Tel: +33(0)3.2087.7793
List of former members from team 5 :
Marie Bicharel Leconte : Engineer
Aurore Hebras : PhD Student / Post doctoral researcher
Cecilia Bellengier : PhD Student